Beyond consultancy, proven to deliver.
Orphan Drug Consultancy provides end-to-end management and technical support to pharmaceutical and biopharmaceutical companies launching new products globally.
The right expertise. Right when you need it.
Our team consists of trusted advisors—industry-leading subject matter experts (SMEs) and professionals—who provide consulting services to innovative pharmaceutical and biotech companies.
Supported over 70+ Pharma and Biopharma Companies
100+ Combined Successful Global Country Launches
Supported over 70+ Pharma and Biopharma Companies
Supported over 70+ Pharma and Biopharma Companies

What our clients say
Swiss Client Event
Swiss Client Event
Vertex QA
Vertex QA
ITF US
ITF US - Matt Tredeau
Fennec
Fennec
Testimonial 5
Testimonial 4
Testimonial 3
Testimonial 1
Title Here
Testimonial 2
Doctor
Case Study
Navigating the landscape of bringing a product to your patient
Navigating the landscape of bringing a product to your patient
Lorem ipsum dolor sit amet consectetur. Massa eu dignissim et mollis ac. Sed felis sed pellentesque eu viverra interdum facilisis sit. Leo lacus pharetra ut tortor condimentum sed mauris a. Suspendisse quam arcu nullam pretium nibh sit elementum duis quisque.
Resources
For Industry Leaders

Pioneering Operational Clarity to Power Growth in Orphan Drugs
Company: Pharming Therapeutics The Project Pharming Therapeutics faced significant challenges due to the absence of a formal Sales & Operations Planning (S&OP) process. Without a structured approach, there was no regular forum for cross-functional collaboration- leaving critical business areas like commercial demand, product launches, regulatory activities, and supply planning uncoordinated. As subject-matter experts, ODC brought […]

Navigating Brexit’s Impact on EU Pharma: Insights from Our Recent Event in Boston
We recently had the pleasure of hosting the “EU Pharma Launch and Brexit: Unanticipated Challenges” event at MassBio in Boston. This gathering brought together industry experts, pharma professionals, and various stakeholders to explore the multifaceted impact of Brexit on EU pharmaceutical product launches. Exploring Crucial Areas The event delved deeply into several critical areas affected […]

Melbourne 2023
Join us in Melbourne for our Masterclass Program and BioForum 14 – 17 Nov 2023 Orphan Drug Consulting will be heading to Melbourne from 14 – 17 November 2023. The team will be delivering a three-part Masterclass Program over four days, including: Part A | BioForum: Tuesday 14 November | 4:00pm – 6:30pm | Allens, […]

CPHI Barcelona 2023
Find us at CPHI Barcelona 2023 24 – 26 Oct Orphan Drug Consulting will be attending CPHI Barcelona 2023 at Fira Barcelona Gran Via from 24th – 26th October 2023. The team is delighted to connect with colleagues across the global pharma community and looking forward to forging new partnerships at CPHI. About CPHI […]

World Orphan Drug Congress Europe 2023
Join us at World Orphan Drug Congress Europe 2023 30 Oct -2 Nov 2023 Orphan Drug Consulting is delighted to be attending World Orphan Drug Congress Europe 2023 in Barcelona at Fira de Barcelona – Montjuïc from 30th October – 2nd November 2023. The team is looking forward to meeting industry leading experts and discussing […]

Swiss Biotech Day 2023
Join us at Swiss Biotech Day 2023 24-25 April 2023. Orphan Drug Consulting is delighted to be attending Swiss Biotech Day 2023 taking place as an event in Basel, Switzerland from 24th–25th April 2023. About Swiss Biotech Day 2023The Swiss Biotech Day 2023 will provide experts from the life sciences sector with the opportunity to […]

World Orphan Drug Congress USA 2023
Join us at World Orphan Drug Congress USA 2023 23-25 May 2023. Orphan Drug Consulting is delighted to be attending World Orphan Drug Congress 2023 taking place as an event in the Gaylord National Resort & Convention Center Washington, D.C. from 23rd–25th May 2023. About World Orphan Drug Congress USA 2023The World Orphan Drug Congress […]

BIO International Convention 2023
Join us at BIO International Convention 2023 5-8 June 2023. Orphan Drug Consulting is excited to be attending BIO International Convention 2023 taking place as an event in Boston, MA from 5th–8th June 2023. About BIO International Convention 2023 BIO represents businesses, universities, state biotechnology centers, and other related organizations throughout the United States and in […]

J.P. Morgan Healthcare Week 2023
January 9th-12th, 2023 CEO and Founder of Orphan Drug Consulting, Evelyn Kelly, will be visiting San Francisco for J.P. Morgan week in January 2023. Widely considered one of the most prominent events in the biotech and pharma industry, Evelyn is excited to meet fellow industry members in San Francisco during this week. Reach out below […]

Orphan Drugs & Rare Diseases Global Congress 2022 Americas
Join us at Orphan Drugs & Rare Diseases Global Congress 2022 Americas November 29-30, 2022. Orphan Drug Consulting is delighted to be attending the Orphan Drugs & Rare Diseases Global Congress 2022 Americas which is taking place as an event in the Hyatt Regency Boston Hotel, MA from 29th–30th November 2022. About Orphan Drugs & […]

European Supply Strategy Workshop
Join us at our Complimentary European Supply Strategy Workshop in Zurich, Switzerland. December 12, 2022. Location: Hotel Schweizerhof Zurich, Bahnhofplatz 7 ,8001 Zurich, Switzerland Date: Monday, 12th December 2022 Workshop Time: 12:00 – 16:30 Social Event Time: 16:30 – 20:00 When preparing for a product launch in Europe, it’s important to choose the […]

Orphan Drug Consulting Celebrates 5 Years in Business with Key Milestone Achievements in 2022
Dublin, Ireland, 28 September 2022 – Orphan Drug Consulting celebrates its fifth anniversary. Orphan Drug Consulting is proud to announce the celebration of 5 years in business. Since its foundation in 2017 by Evelyn Kelly, Orphan Drug Consulting has grown from a core team based in Ireland to a combined global group of 26 employees […]

World Orphan Drug Congress Europe 2022
Join us at World Orphan Drug Congress Europe 2022 November 14-17, 2022. Orphan Drug Consulting is delighted to be exhibiting at World Orphan Drug Congress 2022 taking place as an event in the Melia Sitges Hotel, Spain from 14th–17th November 2022. Join Evelyn Kelly at World Orphan Drug Congress as she presents ‘Navigating the potential […]

Pharma Integrates 2022
Join us at Pharma Integrates 2022 November 17, 2022. The in-person event will take place at the Leonardo Royal Hotel London Tower Bridge in London, United Kingdom. Join Jason Cameron as he presents “The Importance of an Outsourced Manufacturing Strategy and its Alignment with Corporate Goals”. An increasing reliance on virtual/outsourced operations has led to […]

Orphan Drug Consulting Further Expands into the US Market with Key Personnel Relocation to Boston, MA
Dublin, Ireland, 13th September 2022 – Orphan Drug Consulting is delighted to announce the relocation of David Crowley, Head of Client Delivery, to the United States where he will oversee the ongoing expansion of Orphan Drug Consulting’s operations in Boston, MA. David joined the Orphan Drug Consulting team in January 2020 and has been a […]

Orphan Drug Consulting Appoint Mike Gannon as Chief Financial Officer
Dublin, Ireland, 08 September 2022 – Orphan Drug Consulting is delighted to announce the appointment of Mike Gannon as Chief Financial Officer of Orphan Drug Consulting. Mike joined Orphan Drug Consulting in June 2020 as a Consultant specializing in helping client organizations grow and develop internationally, as well as advising on appropriate structures for finance, […]

AusBiotech 2022
Join us at AusBiotech 2022 October 26-28, 2022. Join Orphan Drug Consulting at Australia’s largest life sciences conference at the Perth Convention and Exhibition Centre. This conference is reuniting the biotech community in order to evaluate accomplishments, engage in face-to-face networking, and progress the industry. The program this year offers local and international business leaders […]

CPhI Worldwide 2022
Join us at CPhI Worldwide 2022 November 1-3, 2022. The online and in-person event will take place at the Messe in Frankfurt, Germany. Join us to connect with new partners face-to-face, network, and source innovative pharma solutions on-site from thousands of exhibitors covering the entire pharma supply chain. CPhI Worldwide showcases each stage of the […]

Orphan Drug Consulting Appoint Anne Capplis as Lead Consultant for the Asia Pacific Region
Dublin, Ireland, 24th August 2022 – Orphan Drug Consulting announces the appointment of Anne Capplis as Lead Consultant for the Asia Pacific region to the Orphan Drug Consulting team. Orphan Drug Consulting is delighted to welcome Anne Capplis to its growing international consulting team as Lead Consultant for the Asia Pacific region. Orphan Drug Consulting expanded […]

Orphan Drug Consulting Appoint David McGovern as a Supply Chain Consultant
Dublin, Ireland, 08 August 2022 – Orphan Drug Consulting announces the appointment of David McGovern as a Supply Chain Consultant to the Orphan Drug Consulting team. Orphan Drug Consulting is delighted to welcome David McGovern to its growing international consulting team as a Supply Chain Consultant. David will join Frances Pierce’s team of supply chain […]

Orphan Drug Consulting Appoint Martina White and Nicola Martone as Quality Consultants
Dublin, Ireland, 30 June 2022 – Orphan Drug Consulting announces the appointment of Martina White and Nicola Martone as Quality Consultants to the Orphan Drug Consulting team. Orphan Drug Consulting is delighted to welcome Martina White and Nicola Martone to its growing international consulting team as Quality Consultants. Martina and Nicola will join Sharon Cullen’s team […]

World Orphan Drug Congress USA 2022
Join us at World Orphan Drug Congress USA 2022 July 11-13, 2022. Orphan Drug Consulting is delighted to be exhibiting at World Orphan Drug Congress 2022 taking place as an event in the Hynes Center, Boston, MA, from 11th – 13th July 2022. Find us at booth 526. About World Orphan Drug Congress USA 2022 […]

Almac Product Launch Success Workshop
Join Frances Pierce at Almac’s Product Launch Success Workshop June 21, 2022. Orphan Drug Consulting is delighted to announce that Frances Pierce will be presenting at Almac’s Product Launch Success Workshop in Dublin on the 21st of June. Join Frances Pierce at Almac’s Product Launch Success Workshop as she presents “Trade, custom, and import/export considerations”. […]

Orphan Drug Consulting Appoint Frances Pierce as Head of Supply Chain
Dublin, Ireland, 26 May 2022 – Orphan Drug Consulting announces the appointment of Frances Pierce as Head of Supply Chain on the Orphan Drug Consulting management team. Frances joined Orphan Drug Consulting in July 2021 and has since been promoted to Head of Supply Chain, a key position on Orphan Drug Consulting’s management team. Frances […]

BIO International Convention 2022
Join us at BIO International Convention 2022 June 13-16, 2022. Orphan Drug Consulting is delighted to be attending BIO International Convention 2022 in San Diego, California taking place as an event at the San Diego Convention Center from 13-16 June 2022. About Bio International Convention 2022 BIO International Convention is back in person for the […]

Bio Integrates 2022
Join us at Bio Integrates 2022 May 17, 2022. Orphan Drug Consulting is delighted to be attending Bio Integrates 2022 taking place as an event at the Leonardo Royal Hotel London Tower Bridge on the 17th of May 2022. About Bio Integrates 2022 Life Science Integrates is delighted to announce the fourth Bio Integrates conference, […]

Orphan Drug Consulting CEO and Founder Evelyn Kelly selected for the EY Entrepreneur of the Year Programme 2022
Dublin, Ireland, 05 May 2022 – Orphan Drug Consulting is delighted to announce that CEO and Founder Evelyn Kelly has been selected as a finalist for the EY Entrepreneur of the Year Programme 2022. The EY Entrepreneur of the Year Programme 2022, is a world-class development programme that supports inspires, and connects a community of […]

Product Launch Planning Workshop
Join us at our Product Launch Planning Workshop in Cambridge, MA. May 10, 2022. Orphan Drug Consulting is delighted to invite you to attend our complimentary workshop titled Product Launch Planning – Phase Appropriate Strategies from the Clinic through to Commercial Bringing a pharmaceutical product to the market is no easy task. There are […]

Orphan Drug Consulting Appoint Sharon Cullen as Head of Quality
Orphan Drug Consulting is delighted to welcome Sharon Cullen to its growing international consulting team where she takes up the role of Head of Quality. Sharon Cullen is Head of Quality for Orphan Drug Consulting with extensive experience in the Life Science industry, having previously held roles in Quality Management with a focus on the Pharmaceutical […]

Orphan Drug Consulting Celebrates Four Years in Business
Dublin, Ireland, 21 October 2021 – Orphan Drug Consulting Celebrates its Fourth Anniversary. Orphan Drug Consulting is proud to announce the celebration of 4 years in business. Since its establishment in 2017, Orphan Drug Consulting has grown from a core team based in Ireland to a global group with offices in Dublin, London, and Boston […]

World Orphan Drug Congress 2021
Join us at World Orphan Drug Congress 2021 November 15-18, 2021. Orphan Drug Consulting is delighted to be exhibiting at World Orphan Drug Congress 2021 taking place as an event in Barcelona, Spain from 15th – 18th November 2021. Find us at booth 8. Join Jason Cameron at World Orphan Drug Congress as he presents […]

CPhI Worldwide 2021
Join us at CPhI Worldwide 2021 November 9-11, 2021. The in-person event will take place at the Fiera Milano in Milan, Italy. Join us to connect with new partners face-to-face, network, and source innovative pharma solutions on-site from thousands of exhibitors covering the entire pharma supply chain. CPhI Worldwide showcases each stage of the pharmaceutical […]

Orphan Drug Consulting Appoint Jason Cameron as Chief Operating Officer
Dublin, Ireland, 12 August 2021 – Orphan Drug Consulting is delighted to welcome Jason Cameron to its growing international consulting team where he takes up the role of Chief Operating Officer. Jason has over 25 years of experience in the pharmaceutical industry working across supply chain, distribution, manufacturing, and broader technical operations, with a particular […]

EMA Labelling and Packaging Regulations
Introduction The European Medicines Agency (EMA) is a decentralised agency of the European Union (EU) in charge of scientific evaluation, regulation, and quality control of medicines in the EU. Before approving medicinal products for distribution in the 27 EU member states along with Norway, Lichtenstein, and Iceland, the EMA makes sure that they meet safety, efficiency, and quality standards. In relation to […]

EU Marketing Authorization Procedures
European Medicines Agency The European Medicines Agency (EMA) is responsible for the scientific evaluation of centralized marketing authorization applications (MAA’s). A marketing authorization (MA) is the authorization of a medicine to be sold in one, several, or all the European Union’s member states, depending on the type of application that has been submitted and to which agency. […]

Webinar: Advanced Therapy Conundrum: Should You Build, Outsource, or Go Hybrid?
Webinar: Advanced Therapy Conundrum: Should You Build, Outsource, Or Go Hybrid? June 22, 2021. 2:00PM EST/ 7.00PM GMT Companies developing advanced therapies have to decide early on: Build capacity, and how much? Outsource, and how little? Mix and Match, and how effective? One thing is certain: You need to clearly understand the opportunities and […]

Acting as a Responsible Person (import) (RPI) in the UK
Background The Medicines and Healthcare products Regulatory Agency (MHRA) recently published guidance called “Acting as a Responsible Person (import) (RPi) from 1 January 2021”. The guidance describes the role of an RPi and how to become a RPi. Also of relevance is the guidance “Sourcing medicines for the Great Britain market from an approved country for […]

Webinar: Launching an RX Product in the EU
Webinar: Launching an RX Product in the EU Fail to Prepare, Prepare to Fail [January 27, 2021. 4:00PM GMT/11:00AM EST] How do you manage a successful pharmaceutical product launch with challenges and variables of 27 EU markets? Our webinar will share some of the following key insights: How key differences between EMA and FDA impact […]

BREXIT: Approved Country Lists Post-Transition Period
Countdown to Transition Period End Date Days Hours Minutes Seconds Introduction On 31 December 2020, the Brexit transition period will officially end which means that from 1 January 2021 changes to the current arrangements in place may occur. During the transitional period, the UK has remained a member of the European Economic Area (EEA), the […]

Orphan Drug Consulting Appoint Grainne Higgins as Human Resources Consultant
Dublin, Ireland, 28 September 2020 – Orphan Drug Consulting is delighted to welcome Grainne Higgins to its growing international consulting team. Grainne’s focus will be twofold within Orphan Drug Consulting, both supporting our clients with their Human Resources needs across talent/recruitment/organizational strategy & development; in addition to providing Executive and Leadership coaching to implement best […]

Orphan Drug Consulting Appoint Mike Gannon as Financial and Entity Planning Consultant
Dublin, Ireland, 25 August 2020 – Orphan Drug Consulting is delighted to welcome Mike Gannon to its growing international consulting team. Mike specializes in helping organizations grow and develop internationally as well as advising on optimum structures for finance, tax, treasury and business administration and support functions. Providing this expert advice to Orphan Drug Consulting […]
Orphan Drug Consulting Open Office in Boston, MA, and Appoint Christy Webb as US-based Consultant
Dublin, Ireland, 20 July 2020 – Orphan Drug Consulting today announced the opening of office space in Boston, Massachusetts, and the further growth of its US-based consultant team with the appointment of Christy Webb as a Quality Assurance consultant. This further expansion into the US market by Orphan Drug Consulting addresses an increased demand for […]

Orphan Drug Consulting Appoint Tanya Quinn as US-based Consultant
Dublin, Ireland, 02 June 2020 – Orphan Drug Consulting is delighted to welcome Tanya Quinn (through her company Knowledge Base LLC) to its international consulting team. Tanya will support Orphan Drug Consulting with its growing client requirements in the US from her New York/New Jersey-Metro location. Tanya is a well-respected, experienced senior supply chain management […]
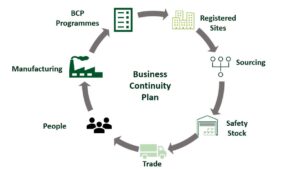
Pharmaceutical Business Continuity Plan as a Key Component of COVID-19 Pandemic Response
“To be a proper business continuity plan, a company needs to roll it out across its entire organization, there needs to be clear roles and responsibilities for people and the plan needs to be simulated and practiced.” In response the COVID-19 pandemic, this webinar examines business continuity planning as a risk mitigation strategy. David […]

EU Regulatory Flexibility During COVID-19 Pandemic
In these uncertain times it can be challenging to comply with regulatory requirements while maintaining supply demands, but there has been advice published by the authorities to help. This guidance has outlined regulatory flexibility that can be applied to help pharmaceutical companies cope with the consequences of the pandemic, while ensuring a high level of […]

EU-GMP Guideline of Annex 21 ‘Importation of Medicinal Products’
On 20th March 2020 the EU-GMP Guideline of Annex 21 ‘Importation of Medicinal Products’ was published as a draft. In addition to the guidance outlined in the main chapters and annexes, it was a thought that there was a need to publish a specific guideline on importation of medicinal products under a dedicated annex of […]

The Impact of Coronavirus (SARS-CoV-2) and COVID-19 on Global Pharmaceutical Supply Chains with Focus on the Indian and Chinese Markets
On 31st of December 2019, a cluster of pneumonia cases with unknown aetiology were reported in Wuhan, China. At the time, healthcare professionals could not identify the causative agent for these seemingly related incidents but on the 7th of January the Chinese authorities confirmed a novel coronavirus (nCoV) was responsible for the disease. This virus […]

Rare Diseases Day 2020: The Importance of Orphan Drugs in Combating Rare Diseases
An Overview of Rare Diseases: On February 29th, 2020 the world will observe Rare Diseases Day coordinated by the European Organisation for Rare Diseases (EURODIS). Now in its 13th year, this special day aims to spread awareness of rare diseases and the patients whose lives are impacted by them. But what exactly is a rare […]

Advanced Therapy Medicinal Products (ATMPs)
With the rise of new therapeutic modalities, regulators have had to adapt to the changing environment and one of these adaptions is through the introduction of Advanced Therapy Medicinal Products (ATMPs). This article highlights some of the key considerations of working with ATMPs.

Brexit
The United Kingdom (UK) voted in a referendum on June 23rd, 2016 to leave the European Union (EU), by 52% to 48%, leaving the Single Market and the Customs Union. While the referendum was not legally binding, the UK government at the time promised to honour the result. The UK has population of over 66 […]

Pioneering Operational Clarity to Power Growth in Orphan Drugs
Company: Pharming Therapeutics The Project Pharming Therapeutics faced significant challenges due to the absence of a formal Sales & Operations Planning (S&OP) process. Without a structured approach, there was no regular forum for cross-functional collaboration- leaving critical business areas like commercial demand, product launches, regulatory activities, and supply planning uncoordinated. As subject-matter experts, ODC brought […]

Navigating Brexit’s Impact on EU Pharma: Insights from Our Recent Event in Boston
We recently had the pleasure of hosting the “EU Pharma Launch and Brexit: Unanticipated Challenges” event at MassBio in Boston. This gathering brought together industry experts, pharma professionals, and various stakeholders to explore the multifaceted impact of Brexit on EU pharmaceutical product launches. Exploring Crucial Areas The event delved deeply into several critical areas affected […]

Melbourne 2023
Join us in Melbourne for our Masterclass Program and BioForum 14 – 17 Nov 2023 Orphan Drug Consulting will be heading to Melbourne from 14 – 17 November 2023. The team will be delivering a three-part Masterclass Program over four days, including: Part A | BioForum: Tuesday 14 November | 4:00pm – 6:30pm | Allens, […]

CPHI Barcelona 2023
Find us at CPHI Barcelona 2023 24 – 26 Oct Orphan Drug Consulting will be attending CPHI Barcelona 2023 at Fira Barcelona Gran Via from 24th – 26th October 2023. The team is delighted to connect with colleagues across the global pharma community and looking forward to forging new partnerships at CPHI. About CPHI […]

World Orphan Drug Congress Europe 2023
Join us at World Orphan Drug Congress Europe 2023 30 Oct -2 Nov 2023 Orphan Drug Consulting is delighted to be attending World Orphan Drug Congress Europe 2023 in Barcelona at Fira de Barcelona – Montjuïc from 30th October – 2nd November 2023. The team is looking forward to meeting industry leading experts and discussing […]

Swiss Biotech Day 2023
Join us at Swiss Biotech Day 2023 24-25 April 2023. Orphan Drug Consulting is delighted to be attending Swiss Biotech Day 2023 taking place as an event in Basel, Switzerland from 24th–25th April 2023. About Swiss Biotech Day 2023The Swiss Biotech Day 2023 will provide experts from the life sciences sector with the opportunity to […]

World Orphan Drug Congress USA 2023
Join us at World Orphan Drug Congress USA 2023 23-25 May 2023. Orphan Drug Consulting is delighted to be attending World Orphan Drug Congress 2023 taking place as an event in the Gaylord National Resort & Convention Center Washington, D.C. from 23rd–25th May 2023. About World Orphan Drug Congress USA 2023The World Orphan Drug Congress […]

BIO International Convention 2023
Join us at BIO International Convention 2023 5-8 June 2023. Orphan Drug Consulting is excited to be attending BIO International Convention 2023 taking place as an event in Boston, MA from 5th–8th June 2023. About BIO International Convention 2023 BIO represents businesses, universities, state biotechnology centers, and other related organizations throughout the United States and in […]

J.P. Morgan Healthcare Week 2023
January 9th-12th, 2023 CEO and Founder of Orphan Drug Consulting, Evelyn Kelly, will be visiting San Francisco for J.P. Morgan week in January 2023. Widely considered one of the most prominent events in the biotech and pharma industry, Evelyn is excited to meet fellow industry members in San Francisco during this week. Reach out below […]

Orphan Drugs & Rare Diseases Global Congress 2022 Americas
Join us at Orphan Drugs & Rare Diseases Global Congress 2022 Americas November 29-30, 2022. Orphan Drug Consulting is delighted to be attending the Orphan Drugs & Rare Diseases Global Congress 2022 Americas which is taking place as an event in the Hyatt Regency Boston Hotel, MA from 29th–30th November 2022. About Orphan Drugs & […]

European Supply Strategy Workshop
Join us at our Complimentary European Supply Strategy Workshop in Zurich, Switzerland. December 12, 2022. Location: Hotel Schweizerhof Zurich, Bahnhofplatz 7 ,8001 Zurich, Switzerland Date: Monday, 12th December 2022 Workshop Time: 12:00 – 16:30 Social Event Time: 16:30 – 20:00 When preparing for a product launch in Europe, it’s important to choose the […]

World Orphan Drug Congress Europe 2022
Join us at World Orphan Drug Congress Europe 2022 November 14-17, 2022. Orphan Drug Consulting is delighted to be exhibiting at World Orphan Drug Congress 2022 taking place as an event in the Melia Sitges Hotel, Spain from 14th–17th November 2022. Join Evelyn Kelly at World Orphan Drug Congress as she presents ‘Navigating the potential […]

Pharma Integrates 2022
Join us at Pharma Integrates 2022 November 17, 2022. The in-person event will take place at the Leonardo Royal Hotel London Tower Bridge in London, United Kingdom. Join Jason Cameron as he presents “The Importance of an Outsourced Manufacturing Strategy and its Alignment with Corporate Goals”. An increasing reliance on virtual/outsourced operations has led to […]

AusBiotech 2022
Join us at AusBiotech 2022 October 26-28, 2022. Join Orphan Drug Consulting at Australia’s largest life sciences conference at the Perth Convention and Exhibition Centre. This conference is reuniting the biotech community in order to evaluate accomplishments, engage in face-to-face networking, and progress the industry. The program this year offers local and international business leaders […]

CPhI Worldwide 2022
Join us at CPhI Worldwide 2022 November 1-3, 2022. The online and in-person event will take place at the Messe in Frankfurt, Germany. Join us to connect with new partners face-to-face, network, and source innovative pharma solutions on-site from thousands of exhibitors covering the entire pharma supply chain. CPhI Worldwide showcases each stage of the […]

World Orphan Drug Congress USA 2022
Join us at World Orphan Drug Congress USA 2022 July 11-13, 2022. Orphan Drug Consulting is delighted to be exhibiting at World Orphan Drug Congress 2022 taking place as an event in the Hynes Center, Boston, MA, from 11th – 13th July 2022. Find us at booth 526. About World Orphan Drug Congress USA 2022 […]

Almac Product Launch Success Workshop
Join Frances Pierce at Almac’s Product Launch Success Workshop June 21, 2022. Orphan Drug Consulting is delighted to announce that Frances Pierce will be presenting at Almac’s Product Launch Success Workshop in Dublin on the 21st of June. Join Frances Pierce at Almac’s Product Launch Success Workshop as she presents “Trade, custom, and import/export considerations”. […]

BIO International Convention 2022
Join us at BIO International Convention 2022 June 13-16, 2022. Orphan Drug Consulting is delighted to be attending BIO International Convention 2022 in San Diego, California taking place as an event at the San Diego Convention Center from 13-16 June 2022. About Bio International Convention 2022 BIO International Convention is back in person for the […]

Bio Integrates 2022
Join us at Bio Integrates 2022 May 17, 2022. Orphan Drug Consulting is delighted to be attending Bio Integrates 2022 taking place as an event at the Leonardo Royal Hotel London Tower Bridge on the 17th of May 2022. About Bio Integrates 2022 Life Science Integrates is delighted to announce the fourth Bio Integrates conference, […]

Product Launch Planning Workshop
Join us at our Product Launch Planning Workshop in Cambridge, MA. May 10, 2022. Orphan Drug Consulting is delighted to invite you to attend our complimentary workshop titled Product Launch Planning – Phase Appropriate Strategies from the Clinic through to Commercial Bringing a pharmaceutical product to the market is no easy task. There are […]

World Orphan Drug Congress 2021
Join us at World Orphan Drug Congress 2021 November 15-18, 2021. Orphan Drug Consulting is delighted to be exhibiting at World Orphan Drug Congress 2021 taking place as an event in Barcelona, Spain from 15th – 18th November 2021. Find us at booth 8. Join Jason Cameron at World Orphan Drug Congress as he presents […]

CPhI Worldwide 2021
Join us at CPhI Worldwide 2021 November 9-11, 2021. The in-person event will take place at the Fiera Milano in Milan, Italy. Join us to connect with new partners face-to-face, network, and source innovative pharma solutions on-site from thousands of exhibitors covering the entire pharma supply chain. CPhI Worldwide showcases each stage of the pharmaceutical […]

Webinar: Advanced Therapy Conundrum: Should You Build, Outsource, or Go Hybrid?
Webinar: Advanced Therapy Conundrum: Should You Build, Outsource, Or Go Hybrid? June 22, 2021. 2:00PM EST/ 7.00PM GMT Companies developing advanced therapies have to decide early on: Build capacity, and how much? Outsource, and how little? Mix and Match, and how effective? One thing is certain: You need to clearly understand the opportunities and […]

Webinar: Launching an RX Product in the EU
Webinar: Launching an RX Product in the EU Fail to Prepare, Prepare to Fail [January 27, 2021. 4:00PM GMT/11:00AM EST] How do you manage a successful pharmaceutical product launch with challenges and variables of 27 EU markets? Our webinar will share some of the following key insights: How key differences between EMA and FDA impact […]
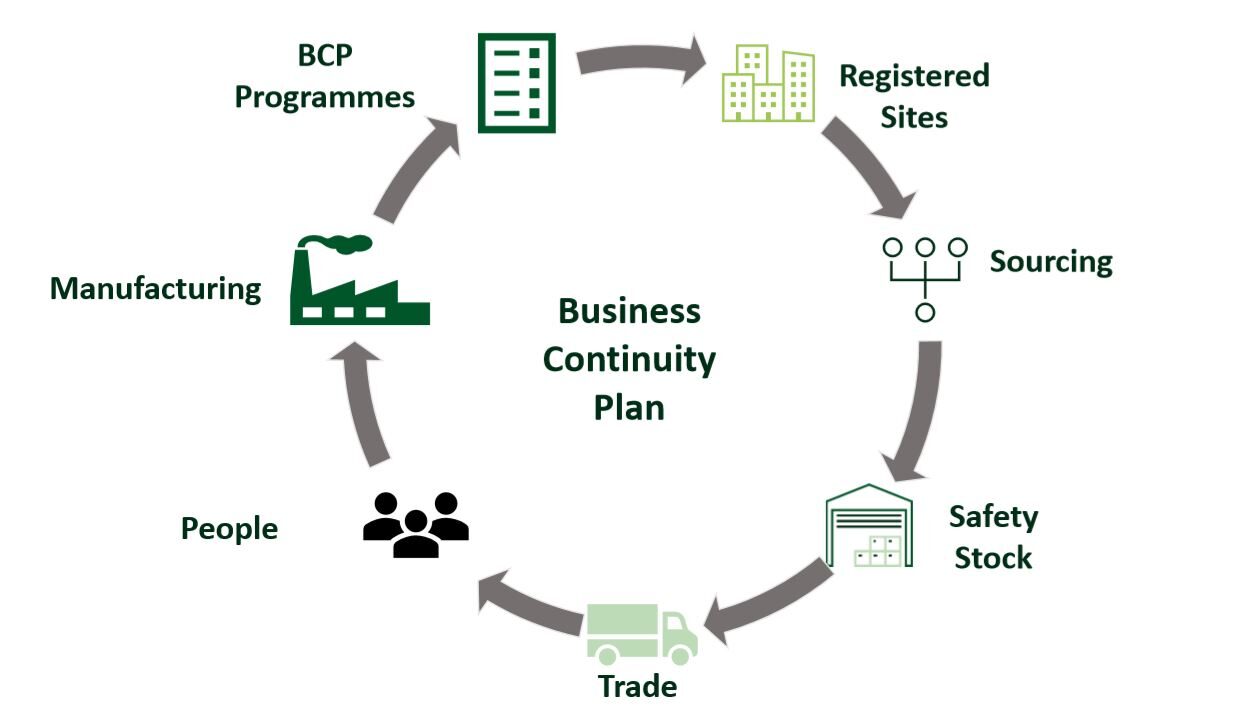
Pharmaceutical Business Continuity Plan as a Key Component of COVID-19 Pandemic Response
“To be a proper business continuity plan, a company needs to roll it out across its entire organization, there needs to be clear roles and responsibilities for people and the plan needs to be simulated and practiced.” In response the COVID-19 pandemic, this webinar examines business continuity planning as a risk mitigation strategy. David […]

Orphan Drug Consulting Celebrates 5 Years in Business with Key Milestone Achievements in 2022
Dublin, Ireland, 28 September 2022 – Orphan Drug Consulting celebrates its fifth anniversary. Orphan Drug Consulting is proud to announce the celebration of 5 years in business. Since its foundation in 2017 by Evelyn Kelly, Orphan Drug Consulting has grown from a core team based in Ireland to a combined global group of 26 employees […]

Orphan Drug Consulting Further Expands into the US Market with Key Personnel Relocation to Boston, MA
Dublin, Ireland, 13th September 2022 – Orphan Drug Consulting is delighted to announce the relocation of David Crowley, Head of Client Delivery, to the United States where he will oversee the ongoing expansion of Orphan Drug Consulting’s operations in Boston, MA. David joined the Orphan Drug Consulting team in January 2020 and has been a […]

Orphan Drug Consulting Appoint Mike Gannon as Chief Financial Officer
Dublin, Ireland, 08 September 2022 – Orphan Drug Consulting is delighted to announce the appointment of Mike Gannon as Chief Financial Officer of Orphan Drug Consulting. Mike joined Orphan Drug Consulting in June 2020 as a Consultant specializing in helping client organizations grow and develop internationally, as well as advising on appropriate structures for finance, […]

Orphan Drug Consulting Appoint Anne Capplis as Lead Consultant for the Asia Pacific Region
Dublin, Ireland, 24th August 2022 – Orphan Drug Consulting announces the appointment of Anne Capplis as Lead Consultant for the Asia Pacific region to the Orphan Drug Consulting team. Orphan Drug Consulting is delighted to welcome Anne Capplis to its growing international consulting team as Lead Consultant for the Asia Pacific region. Orphan Drug Consulting expanded […]

Orphan Drug Consulting Appoint David McGovern as a Supply Chain Consultant
Dublin, Ireland, 08 August 2022 – Orphan Drug Consulting announces the appointment of David McGovern as a Supply Chain Consultant to the Orphan Drug Consulting team. Orphan Drug Consulting is delighted to welcome David McGovern to its growing international consulting team as a Supply Chain Consultant. David will join Frances Pierce’s team of supply chain […]

Orphan Drug Consulting Appoint Martina White and Nicola Martone as Quality Consultants
Dublin, Ireland, 30 June 2022 – Orphan Drug Consulting announces the appointment of Martina White and Nicola Martone as Quality Consultants to the Orphan Drug Consulting team. Orphan Drug Consulting is delighted to welcome Martina White and Nicola Martone to its growing international consulting team as Quality Consultants. Martina and Nicola will join Sharon Cullen’s team […]

Orphan Drug Consulting Appoint Frances Pierce as Head of Supply Chain
Dublin, Ireland, 26 May 2022 – Orphan Drug Consulting announces the appointment of Frances Pierce as Head of Supply Chain on the Orphan Drug Consulting management team. Frances joined Orphan Drug Consulting in July 2021 and has since been promoted to Head of Supply Chain, a key position on Orphan Drug Consulting’s management team. Frances […]

Orphan Drug Consulting CEO and Founder Evelyn Kelly selected for the EY Entrepreneur of the Year Programme 2022
Dublin, Ireland, 05 May 2022 – Orphan Drug Consulting is delighted to announce that CEO and Founder Evelyn Kelly has been selected as a finalist for the EY Entrepreneur of the Year Programme 2022. The EY Entrepreneur of the Year Programme 2022, is a world-class development programme that supports inspires, and connects a community of […]

Orphan Drug Consulting Appoint Sharon Cullen as Head of Quality
Orphan Drug Consulting is delighted to welcome Sharon Cullen to its growing international consulting team where she takes up the role of Head of Quality. Sharon Cullen is Head of Quality for Orphan Drug Consulting with extensive experience in the Life Science industry, having previously held roles in Quality Management with a focus on the Pharmaceutical […]

Orphan Drug Consulting Celebrates Four Years in Business
Dublin, Ireland, 21 October 2021 – Orphan Drug Consulting Celebrates its Fourth Anniversary. Orphan Drug Consulting is proud to announce the celebration of 4 years in business. Since its establishment in 2017, Orphan Drug Consulting has grown from a core team based in Ireland to a global group with offices in Dublin, London, and Boston […]

Orphan Drug Consulting Appoint Jason Cameron as Chief Operating Officer
Dublin, Ireland, 12 August 2021 – Orphan Drug Consulting is delighted to welcome Jason Cameron to its growing international consulting team where he takes up the role of Chief Operating Officer. Jason has over 25 years of experience in the pharmaceutical industry working across supply chain, distribution, manufacturing, and broader technical operations, with a particular […]

EMA Labelling and Packaging Regulations
Introduction The European Medicines Agency (EMA) is a decentralised agency of the European Union (EU) in charge of scientific evaluation, regulation, and quality control of medicines in the EU. Before approving medicinal products for distribution in the 27 EU member states along with Norway, Lichtenstein, and Iceland, the EMA makes sure that they meet safety, efficiency, and quality standards. In relation to […]

EU Marketing Authorization Procedures
European Medicines Agency The European Medicines Agency (EMA) is responsible for the scientific evaluation of centralized marketing authorization applications (MAA’s). A marketing authorization (MA) is the authorization of a medicine to be sold in one, several, or all the European Union’s member states, depending on the type of application that has been submitted and to which agency. […]

Acting as a Responsible Person (import) (RPI) in the UK
Background The Medicines and Healthcare products Regulatory Agency (MHRA) recently published guidance called “Acting as a Responsible Person (import) (RPi) from 1 January 2021”. The guidance describes the role of an RPi and how to become a RPi. Also of relevance is the guidance “Sourcing medicines for the Great Britain market from an approved country for […]

BREXIT: Approved Country Lists Post-Transition Period
Countdown to Transition Period End Date Days Hours Minutes Seconds Introduction On 31 December 2020, the Brexit transition period will officially end which means that from 1 January 2021 changes to the current arrangements in place may occur. During the transitional period, the UK has remained a member of the European Economic Area (EEA), the […]

Orphan Drug Consulting Appoint Grainne Higgins as Human Resources Consultant
Dublin, Ireland, 28 September 2020 – Orphan Drug Consulting is delighted to welcome Grainne Higgins to its growing international consulting team. Grainne’s focus will be twofold within Orphan Drug Consulting, both supporting our clients with their Human Resources needs across talent/recruitment/organizational strategy & development; in addition to providing Executive and Leadership coaching to implement best […]

Orphan Drug Consulting Appoint Mike Gannon as Financial and Entity Planning Consultant
Dublin, Ireland, 25 August 2020 – Orphan Drug Consulting is delighted to welcome Mike Gannon to its growing international consulting team. Mike specializes in helping organizations grow and develop internationally as well as advising on optimum structures for finance, tax, treasury and business administration and support functions. Providing this expert advice to Orphan Drug Consulting […]
Orphan Drug Consulting Open Office in Boston, MA, and Appoint Christy Webb as US-based Consultant
Dublin, Ireland, 20 July 2020 – Orphan Drug Consulting today announced the opening of office space in Boston, Massachusetts, and the further growth of its US-based consultant team with the appointment of Christy Webb as a Quality Assurance consultant. This further expansion into the US market by Orphan Drug Consulting addresses an increased demand for […]

Orphan Drug Consulting Appoint Tanya Quinn as US-based Consultant
Dublin, Ireland, 02 June 2020 – Orphan Drug Consulting is delighted to welcome Tanya Quinn (through her company Knowledge Base LLC) to its international consulting team. Tanya will support Orphan Drug Consulting with its growing client requirements in the US from her New York/New Jersey-Metro location. Tanya is a well-respected, experienced senior supply chain management […]

EU Regulatory Flexibility During COVID-19 Pandemic
In these uncertain times it can be challenging to comply with regulatory requirements while maintaining supply demands, but there has been advice published by the authorities to help. This guidance has outlined regulatory flexibility that can be applied to help pharmaceutical companies cope with the consequences of the pandemic, while ensuring a high level of […]

EU-GMP Guideline of Annex 21 ‘Importation of Medicinal Products’
On 20th March 2020 the EU-GMP Guideline of Annex 21 ‘Importation of Medicinal Products’ was published as a draft. In addition to the guidance outlined in the main chapters and annexes, it was a thought that there was a need to publish a specific guideline on importation of medicinal products under a dedicated annex of […]

The Impact of Coronavirus (SARS-CoV-2) and COVID-19 on Global Pharmaceutical Supply Chains with Focus on the Indian and Chinese Markets
On 31st of December 2019, a cluster of pneumonia cases with unknown aetiology were reported in Wuhan, China. At the time, healthcare professionals could not identify the causative agent for these seemingly related incidents but on the 7th of January the Chinese authorities confirmed a novel coronavirus (nCoV) was responsible for the disease. This virus […]

Rare Diseases Day 2020: The Importance of Orphan Drugs in Combating Rare Diseases
An Overview of Rare Diseases: On February 29th, 2020 the world will observe Rare Diseases Day coordinated by the European Organisation for Rare Diseases (EURODIS). Now in its 13th year, this special day aims to spread awareness of rare diseases and the patients whose lives are impacted by them. But what exactly is a rare […]

Advanced Therapy Medicinal Products (ATMPs)
With the rise of new therapeutic modalities, regulators have had to adapt to the changing environment and one of these adaptions is through the introduction of Advanced Therapy Medicinal Products (ATMPs). This article highlights some of the key considerations of working with ATMPs.

Brexit
The United Kingdom (UK) voted in a referendum on June 23rd, 2016 to leave the European Union (EU), by 52% to 48%, leaving the Single Market and the Customs Union. While the referendum was not legally binding, the UK government at the time promised to honour the result. The UK has population of over 66 […]
